Subscribe to our newsletter!
Follow the latest news from CATALIS and Clinical Trials Quebec

CATALIS has developed a provincial PowerBI dashboard that provides an overview of clinical research in Quebec. Available to research branches, this tool provides standardized data that help follow trends, identify issues, and implement concrete solutions to improve performance. Please contact your research centre if you have any questions.
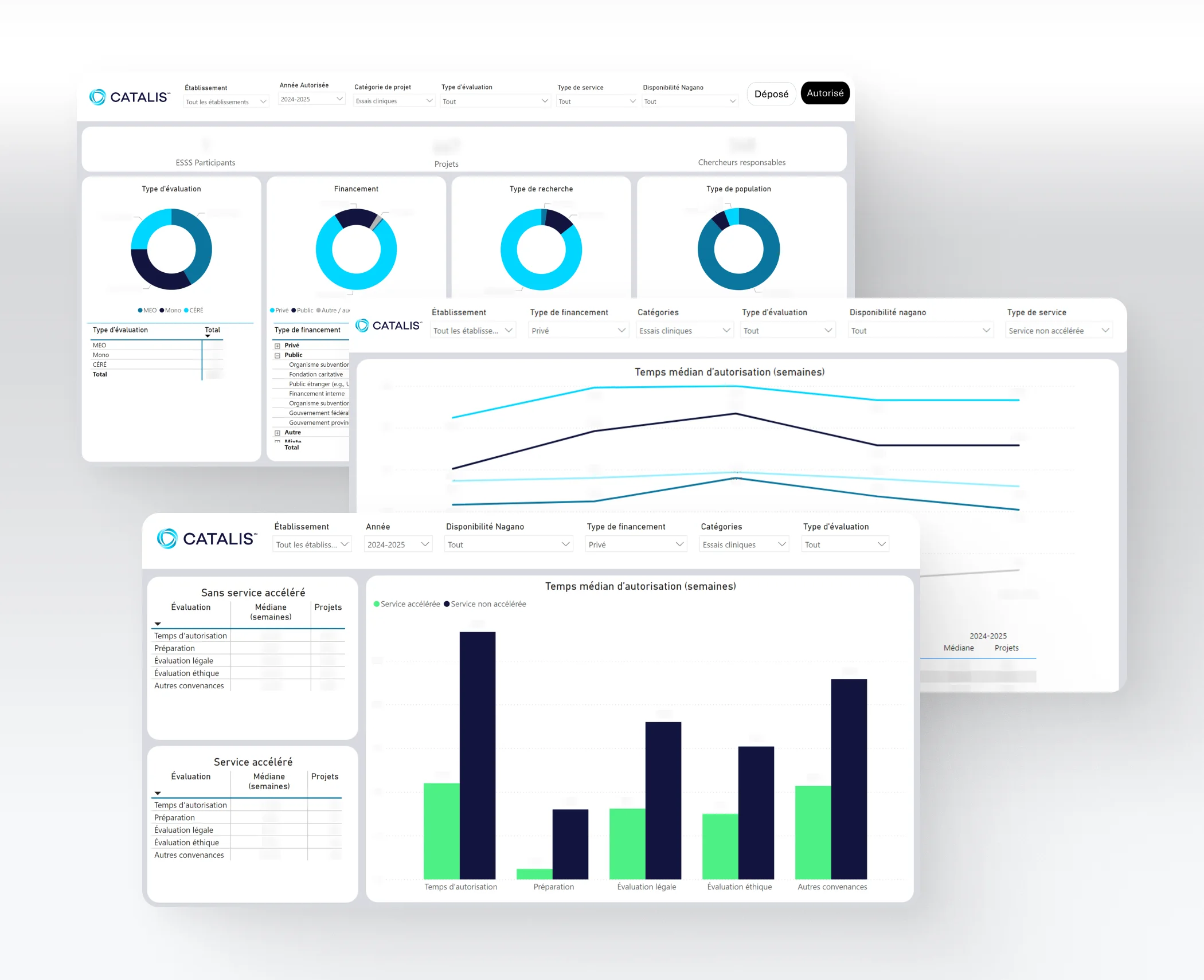
The FAST TRACK Evaluation Service is a unique initiative that positions Quebec as a world leader in clinical research. Its aim is to have clinical trials approved in no more than eight weeks—almost 75% faster than the standard approval time—, thus leaving more time for participant recruitment.
This document provides additional details on the new elements and changes introduced in Revision 3 (R3) of the International Council for Harmonisation (ICH) Good Clinical Practices (GCP).
This memo, developed by the CATALIS Network, clarifies the requirements for signing Form FDA 1572 in Quebec with respect to clinical trials.
The memo, developed by the CATALIS Network, clarifies Canadian and Quebec requirements under Regulation (EU) No. 536/2014with respect to the conduct of clinical trials in Quebec institutions.
The standard clauses include information that must be found in the information and consent forms (ICFs) to comply with the standardization and regulatory requirements in effect in Quebec when carrying out clinical trials. This standardization stems from a consensus of the Research Ethics Boards (REB) of Santé Québec institutions that are members of the CATALIS QUEBEC Network in order to ensure decision-making consistency across the network.
The other harmonized clauses propose common wording used by REBs, in particular in relation to:
Their use in clinical trial consent forms facilitates the drafting of these documents and fast tracks their approval by the institutions part of the health and social services network.
These SOPs intended for REB members and staff are a Quebec adaptation by the CATALIS Network REB of the N2 Canada SOPs. They are reviewed by Quebec’s Ministère de la Santé et des Services sociaux to ensure they meet departmental requirements.
SOPs content:
This statement developed by the CATALIS Network and released by the Ministère de la Santé et des Services sociaux confirms the regulatory requirements applicable to institutions part of the health and social services network, as well as the new provisions of the Charter of the French Language.
The Provincial Model Confidentiality Agreement developed by the CATALIS Network and released by the Ministère de la santé et des services sociaux is recommended to expedite the signing of confidentiality agreements between sponsors and institutions part of the healthcare system. If a sponsor submits this contract in its original form without any modifications, public health institutions in Quebec will accept it as is.
The Pan-Canadian Model Clinical Trial Agreement was developed based on the pan-Canadian model of the Canadian Clinical Trials Coordinating Centre (CCTCC) which the CATALIS Network adapted to the Quebec regulatory context. The provincial mCTA was released by the Ministère de la santé et des services sociaux to expedite the signing of clinical trial agreements between sponsors and institutions part of the healthcare system. If a sponsor submits that mCTA in its original form without any modifications, public health institutions in Quebec will accept it as is.
This document drawn up by the CATALIS Network defines criteria for determining which drugs and procedures can be considered “direct research costs.” Its purpose is to support budget negotiations by providing a shared evaluation framework.
This memo drawn up by the CATALIS Network clarifies the stakeholders’ responsibilities with respect to activities carried out in participants’ homes.
This document drawn up by the CATALIS Network clarifies the impact of the merger of health and social services institutions resulting from the creation of Santé Québec on the negotiation, conclusion and signing of new contracts. It also clarifies the naming of institutions, as approved by Santé Québec, which must be used in all agreements signed since December 1, 2024.
This document is intended to support sponsors and healthcare institutions within Quebec’s health and social services network in the use of software and other technologies in clinical trials. It outlines the applicable responsibilities, clarifies legal and contractual obligations, and provides recommendations to help adapt and standardize contractual practices.
This questionnaire drawn up by the CATALIS Network was designed to expedite the authorization of a research project by providing the research pharmacy with the detailed information needed for its feasibility assessment, in addition to the study documents. It must be completed by the sponsor and submitted by the research teams for evaluation.
Clinical Trials Quebec, powered by CATALIS, provides a directory of Quebec health institutions. It enables users to identify the sites most suitable for carrying out specific clinical trials, thus supporting faster, efficient and collaborative research in service of health.
Quebec’s Public Clinical Trials Database—developed by SemiWeb via the Nagano platform—enables you to find past, ongoing and upcoming clinical trials in Quebec, Canada, and around the world. The Database compiles and shares, in real time, sponsor data from ClinicalTrials.gov (an international public directory) and the data from the Nagano platform taken from public institutions that are part of the Quebec health and social services network.
CATALIS has partnered with N2 Canada to offer CATALIS network member health institutions free access to a shared training platform designed to ensure high-quality research. Through N2 Canada, institutions have access to the CITI-Canada (Collaborative Institutional Training Initiative) training platform.
Please refer to N2 Canada’s comprehensive training offering available to institutions through the partnership with CATALIS.
To access N2 courses, please contact the designated person responsible for providing access at your institution.

These courses jointly created with the private and public members of the CATALIS Quality and Training Advisory Committee address a need expressed by Network members for short, accessible and engaging content to harmonize clinical research practices. They are available for institutions in the N2 digital learning environment. To access these courses, please contact the designated person responsible for providing access to the N2 platform at your institution.
The training courses are only available in French at the moment.

This course presents the fundamentals of research data management. It explains how to distinguish the types of study data and helps to understand the concepts of source and derived data, as well as to ensure data traceability and reconstructibility. The course also covers good document-copying practices and the life cycle of documentation relating to a study.
This course introduces good documentation practices in accordance with ALCOA + principles. Adherence to these principles helps ensure data integrity throughout a study. The course also covers the application of these principles on a day-to-day basis and the proper correction of source documents in the event of errors.
This course is a deep dive into task delegation within a research protocol. It highlights the importance of well-structured delegation to ensure rigour and compliance of clinical studies, and outlines the key steps for organizing the delegation of tasks in accordance with regulatory requirements. These include the identification of tasks that can be delegated, the appropriate selection of appropriate team members, and the rigorous documentation of delegation using adapted logs. The course also covers the management of specific situations, such as changes in personnel or adjustments over the course of the study. Concrete examples and model logs are provided to facilitate implementation.
This course explores the basic principles of quality, compliance and continuous improvement in research.
Presented as a checklist, this tool is intended to guide institutions—particularly their “research office” or equivalent—wishing to undertake research activities involving human participants helping them identify key elements of the administrative and logistical structure needed to guide the research teams.
The purpose of this guide is to help research centres better prepare by sharing proven best practices and avoiding common pitfalls. Designed as a tool kit, it allows you to select relevant information based on your needs or your current stage. It provides a comprehensive and practical resource for navigating all stages of an inspection, from understanding the regulatory framework to taking action after the visit.
Download the checklist and the appendices.
This guide constitutes an introduction to clinical research. It is designed for persons involved in human research, either through direct participation or as administrative or technical support staff. It provides a high-level presentation of the history, different types of studies, key concepts, and the roles of the various stakeholders. It also serves as an entry point to our research program.
This interactive course is intended for researchers involved in clinical research. As a supplement to the “Clinical Research 101” course, it emphasizes the role of researchers and the responsibilities that will enable them to ensure the safety of participants and the regulatory compliance of the studies that they will be conducting.
This course shows the importance of the consent process by covering the related historical and regulatory aspects and its role as a continuous, people-focused process. It then delves into the practical aspects of a meeting aimed at obtaining consent, as well as how specific situations are handled.
Information pending.
Information pending.
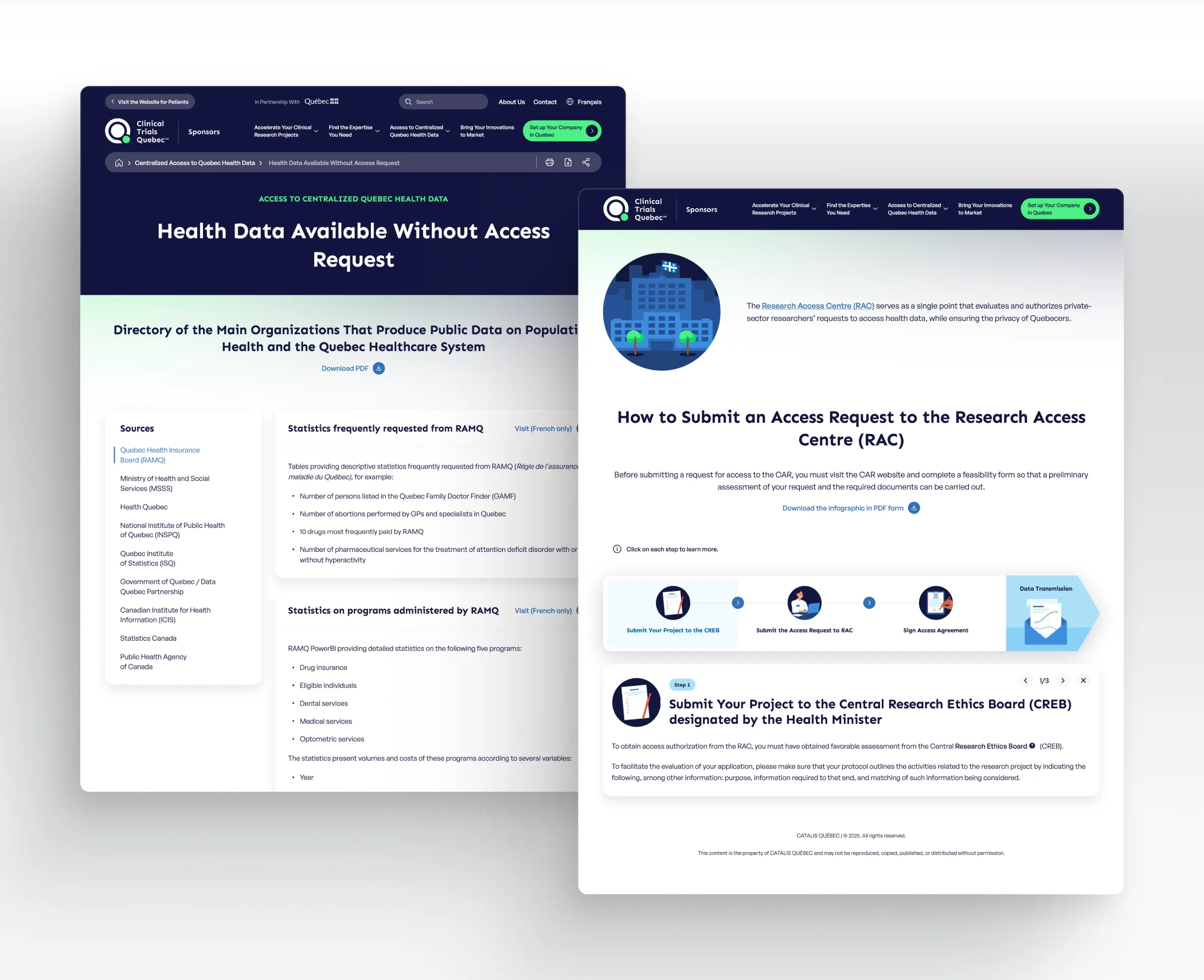
Quebec has an agile and robust system for accessing population health data. Through its universal public healthcare system, Quebec collects massive public health data covering medical procedures, hospitalizations, drugs, and lab or MRI results. Some anonymized health data are accessible to the public, while other data require an access request.
CATALIS’s Personalized Support Service team provides free support to patients, their caregivers and health professionals in searching for clinical trials that meet specified needs and criteria.
