News
Institut de Cardiologie de Montréal recruits the world’s first participant in a Novartis cardiology study
Institut de Cardiologie de Montréal recruits the world’s first participant in a Novartis cardiology study The Institut de Cardiologie de Montréal (ICM – Montreal Heart Institute) has recruited the world’s first participant for the Novartis phase IIa study CPKN605A12201 on atrial fibrillation. Through the FAST TRACK Evaluation Service, and under the leadership of Dr. Laurent Macle and his team,
The CHUS Ranks First in the World for a Sanofi Study on a Rare Inflammatory Disease
The CHUS Ranks First in the World for a Sanofi Study on a Rare Inflammatory Disease The team lead by Dr. Hugues Allard-Chamard at the Centre de recherche du Centre hospitalier universitaire de Sherbrooke (CRCHUS) stood out on the international stage by becoming the first site worldwide to be activated, ahead of 97 other sites in 19 countries, for
New “Clinical Research 101” courses: a comprehensive introduction program for the general public, researchers and research teams
New “Clinical Research 101” courses: a comprehensive introduction program for the general public, researchers and research teams As part of its mandate to strengthen the clinical research ecosystem in Quebec, CATALIS is continuing the progressive roll-out of the Clinical Trials Quebec (CTQ) training program. After launching several tools and capsules for research professionals, we are pleased to present a
Two New Health Institutions Join the CATALIS FAST TRACK Evaluation Service
Two New Health Institutions Join the CATALIS FAST TRACK Evaluation Service The FAST TRACK Evaluation Service continues to grow, and we are proud to announce that two new Health and Social Services Institutions (HSSIs) will soon be offering this Service. The CISSS de la Montérégie-Est and the CIUSSS de la Mauricie-et-du-Centre-du-Québec (CIUSSS MCQ) are one of 21 institutions that have been participating in this initiative to fast track the start of clinical trials in
Good Clinical Practices – ICH E6(R3): Updates to Support the Evolution of Clinical Research
Good Clinical Practices – ICH E6(R3): Updates to Support the Evolution of Clinical Research Clinical research is evolving rapidly, driven by new approaches, emerging technologies, and more flexible trial models. Revision 3 of the Good Clinical Practices (GCP) – ICH E6 fully aligns with this transformation. It provides a modernized, quality‑focused framework that supports more efficient trials while upholding the
Roche Breast Cancer Study: Three Quebec Institutions in the Top 3 Worldwide
Roche Breast Cancer Study: Three Quebec Institutions in the Top 3 Worldwide Thanks to support from each of the FAST TRACK Evaluation Service’s stakeholders, three Quebec hospitals stood out on the international stage for the speed with which they implemented the second phase of Roche’s neoTOV breast cancer study. The Jewish General Hospital (JGH), with Dr. Stephanie Wong, was
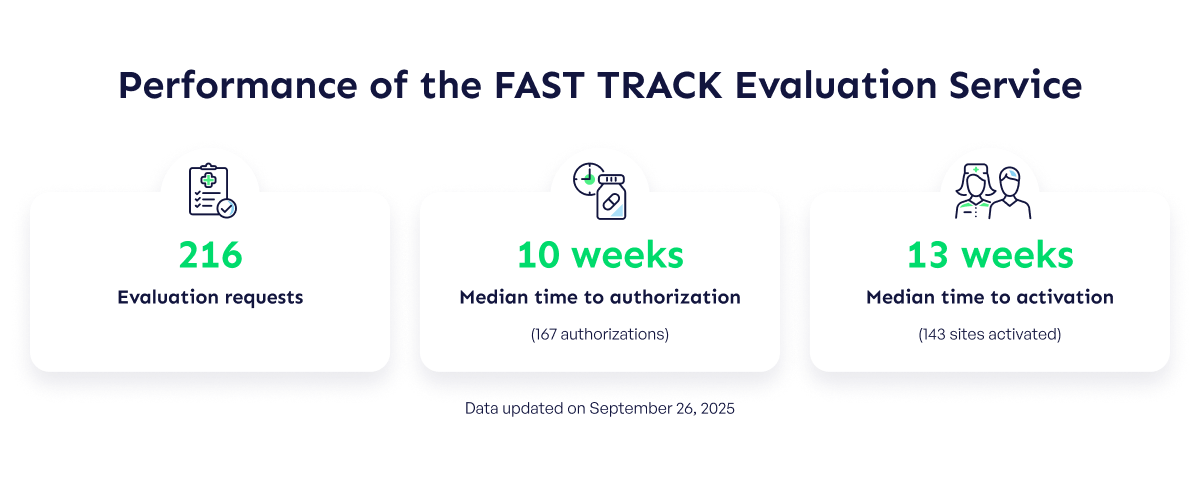
CATALIS Celebrates Four Years of Success With the FAST TRACK Evaluation Service
CATALIS Celebrates Four Years of Success With the FAST TRACK Evaluation Service CATALIS is proud to celebrate four years of success for the FAST TRACK Evaluation Service, which continues to demonstrate its effectiveness in optimizing administrative processes in clinical research in Quebec. Since the launch of the pilot project in 2021, 216 evaluation requests have been processed by the
Montreal’s Jewish General Hospital Authorizes a Novartis Study on GEP-NETs in Only 8 Weeks and Becomes the Second Site Activated Worldwide
Montreal’s Jewish General Hospital Authorizes a Novartis Study on GEP-NETs in Only 8 Weeks and Becomes the Second Site Activated Worldwide The Jewish General Hospital (JGH) in Montreal authorized Novartis’ NETTER-3 study in only 8 weeks with support from the FAST TRACK Evaluation Service. It was fully activated in only 10.2 weeks and allowed the team led by Dr.
CATALIS Unveils Its Clinical Trials Quebec Training Program
CATALIS Unveils Its Clinical Trials Quebec Training Program As part of our 2025–2026 action plan, CATALIS was tasked with promoting best practices in clinical research in Quebec by implementing a provincial training program for research professionals. On one hand, following recommendations from our Network, we renewed our partnership with N2 Network of Networks to provide Québec healthcare institutions with
CATALIS Honors Louise Proulx at the Conclusion of Her Term on the Board of Directors
CATALIS Honors Louise Proulx at the Conclusion of Her Term on the Board of Directors CATALIS wishes to express its gratitude and acknowledge the end of Ms. Louise Proulx’s term as a member of the Board of Directors and Chair of the Audit and Risk Management Committee. An active member of the Board since 2018, Ms. Proulx has played a key role in